NON-SMALL CELL LUNG CANCER (NSCLC)
- 27 Jul, 2015
- Written by Team Dr Lal PathLabs
Medically Approved by Dr. Shuchi
Table of Contents
INTRODUCTION
Lung cancer is the leading cause of cancer-related deaths. Histologically, the World Health Organization (WHO) classifies lung cancer into Non-Small Cell Lung Cancer (NSCLC, 85%) and Small-Cell Lung Cancer (SCLC, 15%). NSCLC is further divided into three major subgroups: Adenocarcinoma, Squamous cell carcinoma and a less well-characterized group of Large cell carcinoma (40%, 30% and 15% of all lung cancer cases respectively). Based on the patient’s medical status and stage of disease, there are four general treatment strategies which includes surgery, radiation therapy, chemotherapy and/or targeted therapy.
Further, tumor subtyping is now essential to appropriately select therapy and determine molecular testing strategies.
Increasingly, the incorporation of molecular testing results is an essential component of clinical decision making in NSCLC.
They form the backbone of personalized medicine, which in the not too distant future will be how all NSCLC patients are treated.
INDIAN SCENARIO
• Lung cancer is a highly aggressive malignancy causing high morbidity and mortality
• Non-Small Cell Lung Carcinoma (NSCLC) accounts for 80-85% of all lung carcinomas and adenocarcinoma is the predominant histologic type
• An increasing incidence of lung cancer has been observed in India
• One million of the current 5 million deaths in the world and 2.41 million in the developing countries is contributed by India and in 2020, this figure is projected at 1.5 million
MOLECULAR BIOMARKERS
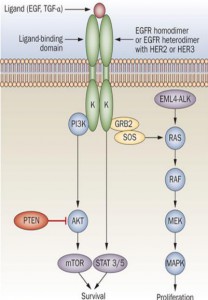
Molecular alterations that occur during lung carcinogenesis or any other cancer result in dysregulation of signaling pathway. The major genes targeted in cancer are the proto-oncogenes and tumor suppressor genes. The various oncogenes and tumor suppressors that are altered in lung cancer and their role in cell signaling pathways (Figure 1) explain their molecular contributions in carcinogenesis.
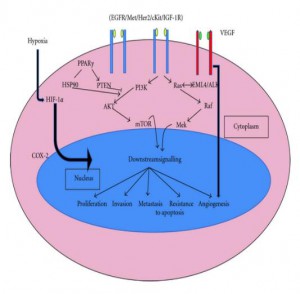 Figure 1: Major signaling pathways affected by altered genes in lung cancer
Figure 1: Major signaling pathways affected by altered genes in lung cancer
CLINICAL SIGNIFICANCE OF MOLECULAR BIOMARKERS
Though any one of the subtypes of lung cancer (SCLC or NSCLC) may be more susceptible to a certain type of mutation, many of the gene alterations that occur in lung cancer are common to both Small-Cell Lung Cancer (SCLC) and Non-Small Cell Lung Cancer (NSCLC). The major driver mutations in lung adenocarcinoma and squamous cell carcinoma are represented in Figures 2a and 2b.
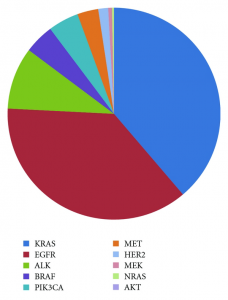 Figure 2a: Mutation spectra in lung adenocarcinoma.
Figure 2a: Mutation spectra in lung adenocarcinoma.
 Figure 2b: Mutation spectra in squamous cell carcinoma.
Figure 2b: Mutation spectra in squamous cell carcinoma.
- EGFR GENE MUTATIONS
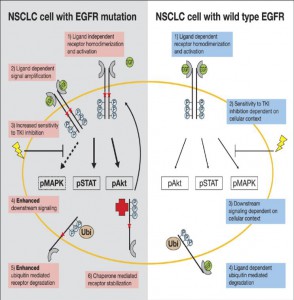
• Epidermal Growth Factor Receptor (EGFR) gene is defective in about 10% of NSCLC patients (and nearly 50% of lung cancer cases in people who never smoked)
• Mutations in four exons18-21 of EGFR gene have been reported to respond to selective anti-EGFR tyrosine kinase inhibitors
• There is a significant first-line benefit with treatment with EGFR TKIs compared with traditional combined platinum-based chemotherapy in patients with tumors harboring an EGFR mutation
• The three most commonly utilized targeted agents against EGFR mutations are gefitinib, erlotinib and cetuximab
- ALK GENE MUTATION
• A rearrangement in the Anaplastic Lymphoma Kinase: ALK gene (most commonly resulting in an EML4-ALK fusion gene) is present in approximately 5% of NSCLC and is more common in non-smokers
• Identification of patients with this rearrangement is of key importance owing to the availability of crizotinib, a newly approved targeted therapy with activity against the kinases of the products of ALK, ROS1 and MET genes
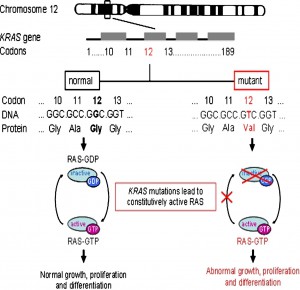
- KRAS GENE MUTATIONS
• Kirsten rat sarcoma viral oncogene, or v-Ki-ras-2 (KRAS) are present in approximately 30% of pulmonary adenocarcinomas and 5% of pulmonary squamous cell carcinomas
• In contrast to the tendency for EGFR gene mutations and EML4-AKL fusion to occur in those who have never smoked, KRAS mutations are more common in past or current smokers
• The majority of the time the identification of a KRAS mutation is a strong negative predictor for identifying an additional aberration in either EGFR or ALK
• Because it is commonly present overall in NSCLC, one potential role for KRAS testing is as a “first-line screening” test, that allows for exclusion of other testing, if positive
OTHER EMERGING MOLECULAR MARKERS
Additional biomarkers currently being studied for application in cancer treatment include the PIK3CA, HER2, BRAF, ROS, RET, NRAS, MET and MEK1 biomarkers. As research continues, more biomarkers are being discovered, each having the potential to open up treatment options that might not have existed previously.
IN BRIEF
Molecular characterization of lung carcinoma contributes valuable information in terms of the patient’s diagnosis, prognosis and the potential for treatment with targeted therapy.
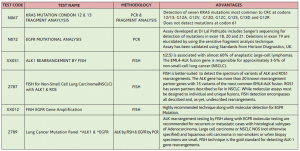
KEY-TESTS RANGE AVAILABLE ON LUNG CANCER
1. Thankappan KR & Thresia CU. Tobacco use & social status in Kerala .Indian J Med Res 2007
2. Gupta R. Smoking , Educational status and health inequity in India. Indian J Med Res 2006
3. Gandara DR, Li T, Lara PR Jr, et al. Algorithm for codevelopment of new drug-predictive biomarker combinations: accounting for inter- and intrapatient tumor heterogeneity. Clin Lung Cancer. 2012
4. Riely GJ, Marks J, Pao W. KRAS mutations non-small cell lung cancer. Proc Am Thorac Soc. 2009
5. Rosell R, Moran T, Queralt C, et al. Screening for epidermal growth factor receptor mutations in Lung Cancer. N Engl J Med. 2009
6. Chiarle R, Voena C, Ambrogio C, Piva R, Inghirami G. The anaplastic lymphoma kinase in the pathogenesis of cancer [review]. Nat Rev Cancer. 2008;8:11-23.
7. National Institutes of Health. ClinicalTrials.gov. www.clinicaltrials.gov. Accessed September 21, 2011.
8. Chougule A, Prabhash K, Noronha V, Joshi A, Thavamani A, et al. (2013) Frequency of EGFR Mutations in 907 Lung Adenocarcioma Patients of Indian Ethnicity. PLoS ONE 8(10).
9. Sasaki T, Rodig SJ, Chirieac LR, et al. The biology and treatment of EML4-ALK non-small cell lung cancer. Eur J Cancer. 2010
10. Yasuda H, de Figueiredo-Pontes LL, Kobayashi S, et al. Preclinical rationale for use of the clinically available multitargeted tyrosine kinase inhibitor crizotinib in ROS1- translocated lung cancer. J Thorac Oncol. 2012
11. Roberts PJ, Stinchcombe TE, Der CJ, et al. Personalized medicine in non-small-cell lung cancer: is KRAS a useful marker in selecting patients for epidermal growth factorreceptor-targeted therapy? J Clin Oncol. 2010
12. Ettinger DS, Akerley W, Bepler G, et al. Non-small cell lung cancer. J Natl Compr Canc Netw.2010;8:740-801.
 National Customer Care : Tel: 011-3988-5050
National Customer Care : Tel: 011-3988-5050
Corporate Office: 12th Floor, Tower B, SAS Tower, Medicity,
Sec- 38, Gurgaon- 122 001, Haryana
Tel: 0124- 3016 500 | Fax: 0124- 42344668
Main Lab: Eskay House, 54, Hanuman Road, New Delhi-110001
www.lalpathlabs.com
doctorfeedback@lalpathlabs.com