Role of Active Vitamin B12 in Human Body
- 13 Apr, 2016
- Written by Team Dr Lal PathLabs
Medically Approved by Dr. Shuchi
Table of Contents
ACTIVE B12
(HOLOTRANSCOBALAMIN)
Studies have shown that active Vitamin B12 deficiency is highly prevalent, especially in certain populations. Elderly persons, people with certain gastrointestinal disorders or people with low dietary intake of Vitamin B12, are at a high risk for developing a deficiency [1– 5]. Severe Vitamin B12 deficiency can lead to serious irreversible neurological damages. Thus, early diagnosis is beneficial and accurate methods for detecting Vitamin B12 deficiency are required.
Vitamin B12 is important for DNA synthesis, formation & maintenance of myelin sheaths, the synthesis of neurotransmitters, erythropoiesis and maintenance of the methyl groups transfers required for numerous methylation reactions such as DNA-methylation. Clinical vitamin B12 deficiency includes two main manifestations: Hematological and/or Neuropsychiatric disorders. Symptoms can develop when the total Vitamin B12 is still within the laboratory reference range, therefore total Vitamin B12 level in the blood may not be indicative of true Vitamin B12 deficiency [2,6].Vitamin B12 and / or folate deficiency may cause macrocytosis which can be masked by concomitant iron deficiency or other disorders. Compared to available modern laboratory markers (like Active B12 [Holotranscobalamin]), macrocytic anemia is regarded as a late and unspecific indicator of Vitamin B12 deficiency.
Often total Vitamin B12 concentrations of less than 150 pmol/L are considered indicative of deficiency.
However:
- A proportion of individuals with low total Vitamin B12 levels have no clinical or biochemical evidence of deficiency
- Neuropsychiatric and metabolic abnormalities can occur with plasma total Vitamin B12 concentrations being well within the normal reference interval
- Reference intervals can vary quite markedly between laboratories
- Large indeterminate zones are being reported
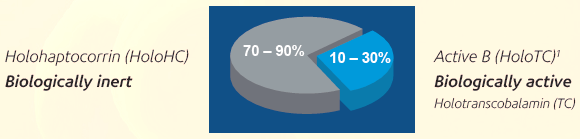
Intrinsic Factor (IF), Transcobalamin (TC) and Haptocorrin (HC) are binding proteins involved in the assimilation, transport and delivery of Vitamin B12 to the cells. When Vitamin B12 is bound to Transcobalamin and Haptocorrin, the resulting complexes are known as Holotranscobalamin (HoloTC, Active B12) and Holohaptocorrin (HoloHC) respectively. Only HoloTC is considered to be bioavailable because it can be taken up by the cells via a specific receptor, the TC receptor. HoloTC is a minor fraction of the total Vitamin B12 in the blood and it varies between 10% and 30 % of total Vitamin B12. Most of the Vitamin B12 in blood (70 – 90 %) is bound to HC. The function of HoloHC is not known.
In recent years, new and sensitive diagnostic markers to determine a person’s Vitamin B12 status have become available. The measurement of total Vitamin B12 in serum has limited sensitivity and specificity for Vitamin B12 deficiency, especially when total vitamin B12 concentrations are less than 400 pmol/L. Measuring total Vitamin B12 can underestimate Vitamin B12 deficiency because the portion of Holotranscobalamin (HoloTC) within the total Vitamin B12 is not linear over the whole serum Vitamin B12 concentration range. This means that total Vitamin B12 concentrations may be within the reference range but the concentration of HoloTC may be low. Since HoloTC is the active form of the vitamin, low HoloTC can cause clinical manifestations of Vitamin B12 deficiency even when the total vitamin B12 is normal [2]. Total Vitamin B12 concentration, that is at the lower end of the reference range may not exclude low HoloTC and thus Vitamin B12 deficiency cannot be excluded [3, 6, 7]. Therefore, Active B12 (HoloTC), is the earliest laboratory marker for Vitamin B12 depletion. Low Active B12 (HoloTC) indicates that the active form of Vitamin B12 is not available, while elevated Methylmalonic Acid (MMA) indicates that Vitamin B12-dependent reactions are impaired. MMA represents a functional marker for Vitamin B12 deficiency. Isolated lowering of Active B12 (HoloTC) shows that the bioavailable Vitamin B12 is depleted (negative B12 balance), while lowered Active B12 (HoloTC) plus elevated MMA (and homocysteine) indicate a metabolically manifested Vitamin B12 deficiency [4, 6, 8]. At this stage, clinical symptoms may be completely absent. However, there is no universal marker for vitamin B12 status for all cases. There are some limitations for each available marker in certain group of patients such as renal patients. However, a combination of markers in these cases can be very helpful in determining the diagnosis and monitoring patients after starting the treatment.
Active B12 (HoloTC) has a shorter biological half-life than HoloHC (12 hrs. vs 9 –10 days [9] ). Thus, concentrations of Active B12 (HoloTC) in plasma reflect very recent changes in Vitamin B12 status (depletion or repletion). Active B12 (HoloTC) has been reported to be low in patients with biochemical signs of Vitamin B12 deficiency such as elevated MMA and homocysteine [10 –12]. This is a common finding in patients with renal diseases, diabetics, elderly subjects or people adhering to a vegetarian diet. Patients with neurological symptoms of unknown etiology should be tested for Vitamin B12 deficiency. A low intake of Vitamin B12, malabsorption, pernicious anemia and gastrointestinal disorders with a shift in pH should be considered in the diagnosis and treatment of Vitamin B12 deficiency.
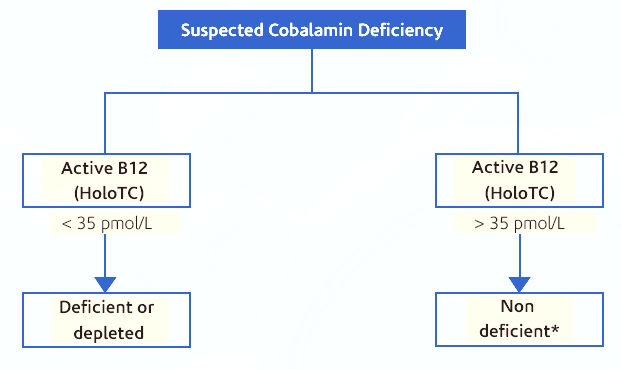
As the first clinical manifestations of Vitamin B12 deficiency are nonspecific, those at risk should have their B12 status checked regularly, every two to three years. Low plasma Active B12 (HoloTC) reflects a depletion status or a deficiency. The increase in Active B12 (HoloTC) after an oral Vitamin B12 supplementation is the earliest indicator that Vitamin B12 is being absorbed. Active B12 (HoloTC) will increase before any change in total B12 or MMA can be recognized [13]. This marker is therefore very promising for early detection of Vitamin B12 deficiency and testing the benefit from oral vitamin B12 treatment. It has been shown in randomized studies that oral Vitamin B12 substitution in persons with normal absorption is effective and improves neurological and haematological symptoms [14–16]. The diagnostic use of Active B12 may allow for the early diagnosis and treatment before irreversible neurological damage occurs. Figure 1 provides the suggested test algorithm for diagnosing the Vitamin B12 status.
- Miller, J.W.; Garrod, M.G.; Allen, L.H.; Haan, M.N. and Green, R. (2009) Am. J Clin Nutr., 90(6), 1586 –1592.
- Lee, Y.K.; Kim, H.S. and Kang, H.J. (2009) Ann. Clin Lab Sci, 39(4), 361– 366.
- Clarke, R.; Sherliker, P.; Hin, H.; Nexo, E.; Hvas, A.M.; Schneede, J.; Birks, J.; Ueland, P.M.; Emmens, K.; Scott, J.M. et al. (2007) Clin Chem, 53(5), 963 – 970.
- Herrmann, W.; Obeid, R.; Schorr, H. and Geisel, J. (2005) Curr. Drug Metab, 6(1), 47– 53.
- Obeid, R.; Schorr, H.; Eckert, R. and Herrmann, W. (2004) Clin Chem, 50(1), 238 – 241.
- Obeid, R. and Herrmann, W. (2007) Clin Chem Lab Med, 45(12), 1746 –1750. [7] Carmel, R. (2011) Am. J Clin Nutr., 94(1), 348S – 358S.
- Herrmann, W.; Schorr, H.; Obeid, R. and Geisel, J. (2003) Am. J. Clin Nutr., 78(1), 131–136.
- Hom, B.L. (1967) Scand J Haematol., 4(5), 321– 332.
- Nexo, E. and Hoffmann-Lucke, E. (2011) Am. J Clin Nutr., 94(1), 359S – 365S [11] Lindgren, A.; Kilander, A.; Bagge, E. and Nexo, E. (1999) Eur. J. Clin. Invest, 29(4), 321– 329.
- Herrmann, W. and Obeid R (2009) European Haematology, 3(1), 7–11.
- Bor, M.V.; Nexo, E. and Hvas, A.M. (2004) Clin Chem, 50(6), 1043 –1049.
- Andres, E.; Vidal-Alaball, J.; Federici, L.; Loukili, N.H.; Zimmer, J. and Kaltenbach, G. (2007) Eur J Intern. Med, 18(6), 456 – 462.
- Andres, E.; Kurtz, J.E.; Perrin, A.E.; Maloisel, F.; Demangeat, C.; Goichot, B. and Schlienger, J.L. (2001) Am. J. Med, 111(2), 126 –129.
- Kuzminski, A.M.; Del Giacco, E.J.; Allen, R.H.; Stabler, S.P. and Linden- baum, J. (1998) Blood, 92(4), 1191–1198.
- Herrmann W, Obeid R in “Labor und Diagnose” (Editor L. Thomas), edition 8, Chapter 13, TH-Books Verlagsgesellschaft-mbH, Frankfurt/Main, in press