What Doctors Need To Know About MDR-TB
- 12 Apr, 2018
- Written by Team Dr Lal PathLabs
Medically Approved by Dr. Shuchi
Table of Contents
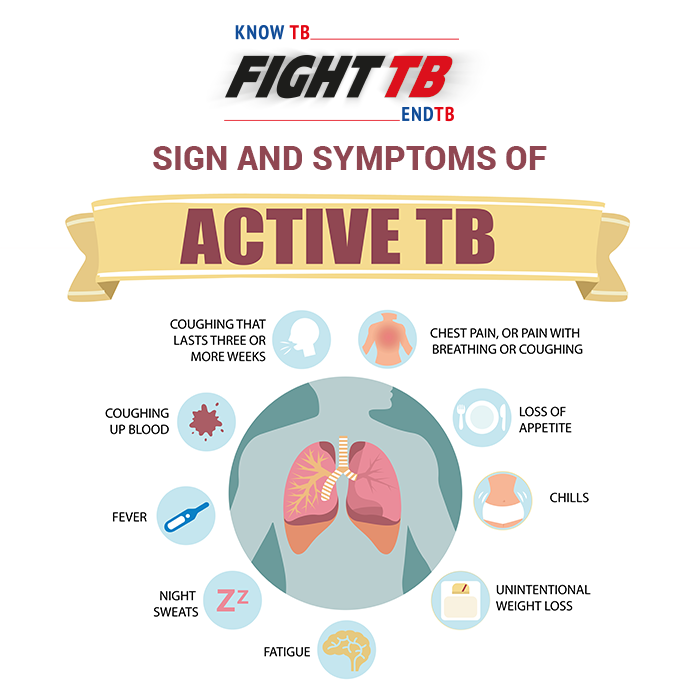
According to WHO, an estimated 490,000 people worldwide developed MDR-TB in 2016, and an additional 110,000 people contracted rifampicin-resistant Tuberculosis . This is the real challenge in the management and treatment of TB.
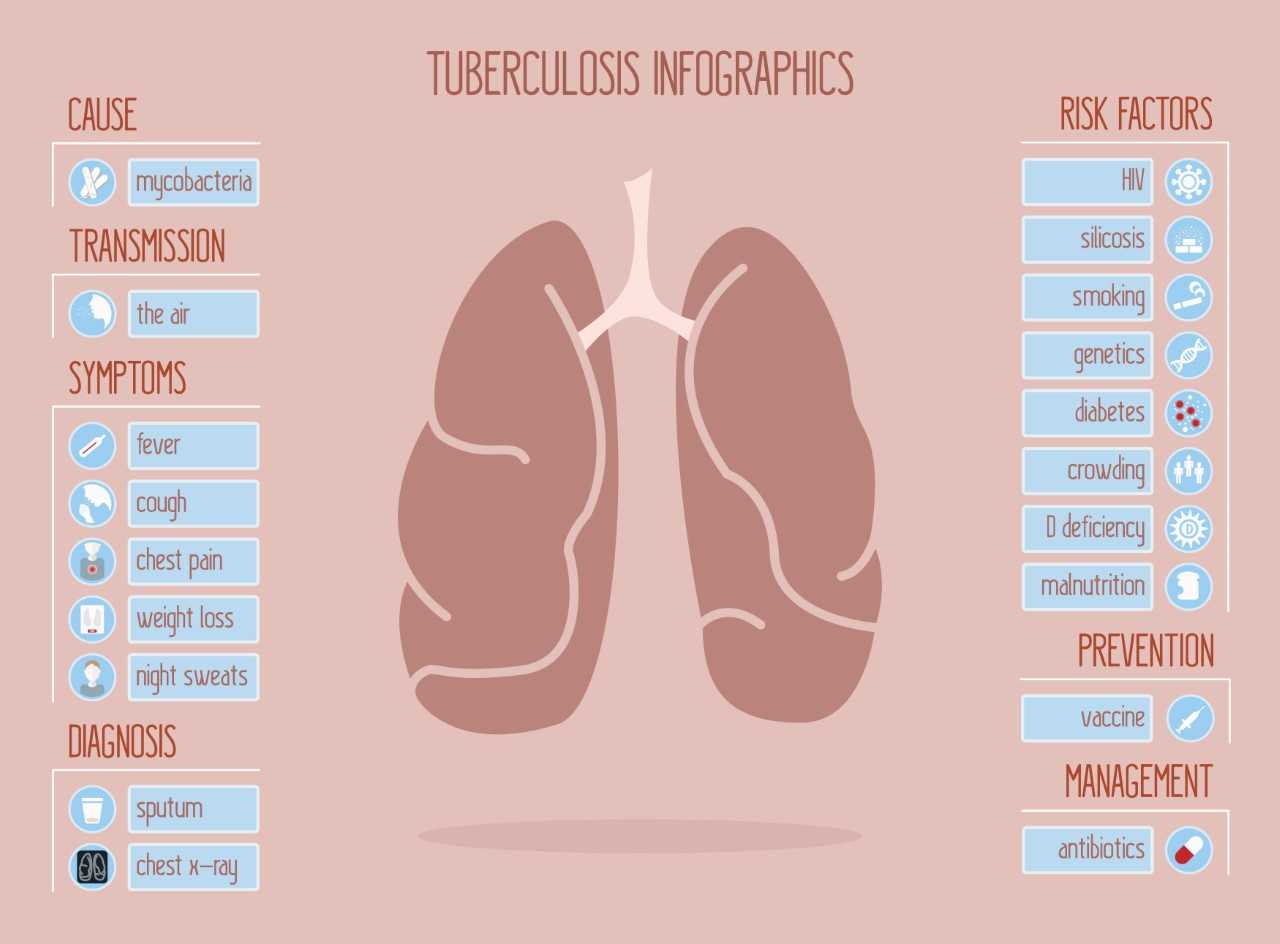
Multidrug-resistant tuberculosis (MDR-TB) is a type of Tuberculosis that does not respond to anti-TB drugs, such as isoniazid and rifampin, the two most powerful ones. This occurs when Mycobacterium tuberculosis (TB-causing bacteria) develops resistance to the antimicrobial drugs.
Emergence and spread of MDR-TB
The main reason for the emergence and spread of MDR-TB is the mismanagement of Tuberculosis treatment arising from:
- Inappropriate or incorrect use of antimicrobial drugs
- Use of ineffective formulations of drugs ( e.g. single drug formulation, medicines of poor quality or inappropriate storage conditions)
- Early treatment interruption or cessation
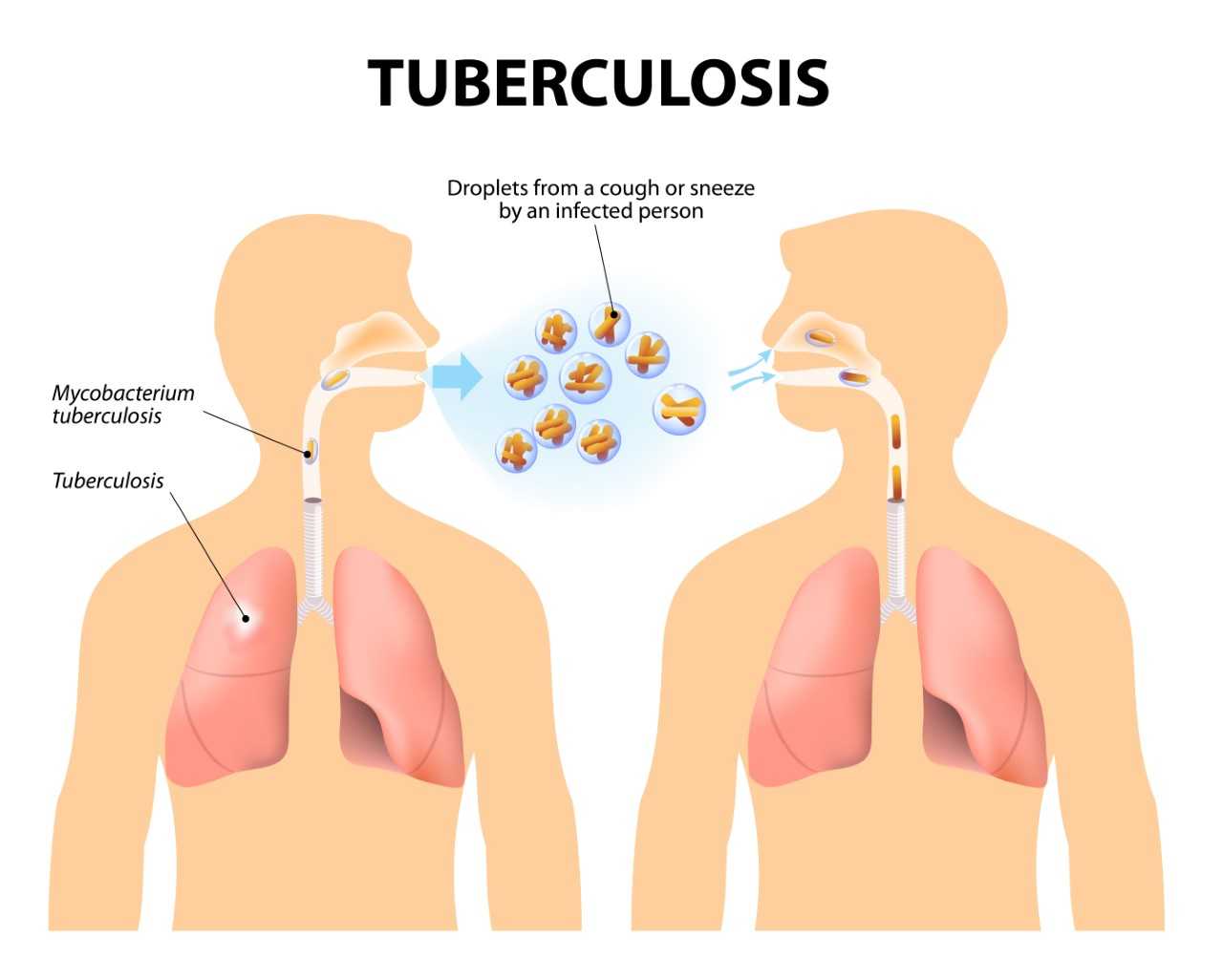
It can also be transmitted, especially in crowded settings such as prisons and hospitals.
Challenges of managing MDR-TB
Limited and expensive medication is the biggest challenge in managing and treating MDR-TB. Often, patients experience many adverse effects from the drugs and tend to stop taking them in between, further decreasing their chances of survival. In some cases, extensively drug-resistant TB (XDR-TB) may develop with additional resistance to more anti-TB drugs, and this responds to even fewer available medicines. So, to prevent these challenges, it is best to control drug-resistant TB by:
- Curing every TB case the first time around
- Providing immediate access to diagnosis for early management
- Ensuring adequate infection control in hospitals to prevent spread
- Ensuring appropriate use of recommended second-line drugs
Diagnosis of MDR-TB
Special laboratory tests such as Xpert MTB/RIF (molecular in type), TB culture test, and TrueNat test help in identifying the disease. These test the bacteria for sensitivity to the drugs or detect resistance patterns. Molecular testing is advantageous as it can provide results within hours.
Management of MDR-TB
MDR-TB is treated with second-line drugs, usually four or more anti-TB drugs. The minimum duration of the treatment is six months. In case of identified rifampin resistance in the specific strain of TB with which the patient is infected, the duration could be extended to 18-24 months. MDR-TB may also require surgery.
Preferred agents used to treat MDR-TB include fluoroquinolones (e.g., moxifloxacin and levofloxacin) and aminoglycosides (amikacin, capreomycin, and kanamycin). In addition, other agents such as linezolid, amoxicillin/clavulanate, clarithromycin, and imipenem may also be used. According to WHO guidelines, the following five-agent treatment regimen should be given for MDR-TB:
- pyrazinamide;
- a fluoroquinolone;
- a parenteral agent (typically amikacin or kanamycin);
- ethionamide (or prothionamide); and
- either cycloserine or para-aminosalicylic acid, with the preference for cycloserine
New developments in MDR-TB management
New WHO recommendations for management of MDR-TB aim at speeding up disease detection, and simultaneously improving its treatment outcomes by using the novel rapid diagnostic test and a shorter, cheaper treatment regimen. The new treatment regimen is less expensive and can be completed in 9-12 months. It is also expected to improve outcomes and potentially decrease deaths due to better adherence and reduced loss to follow-up.