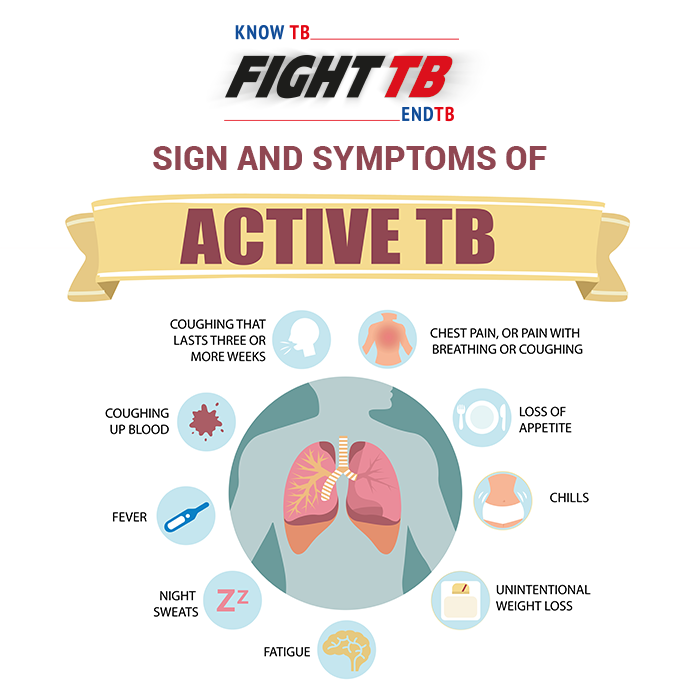
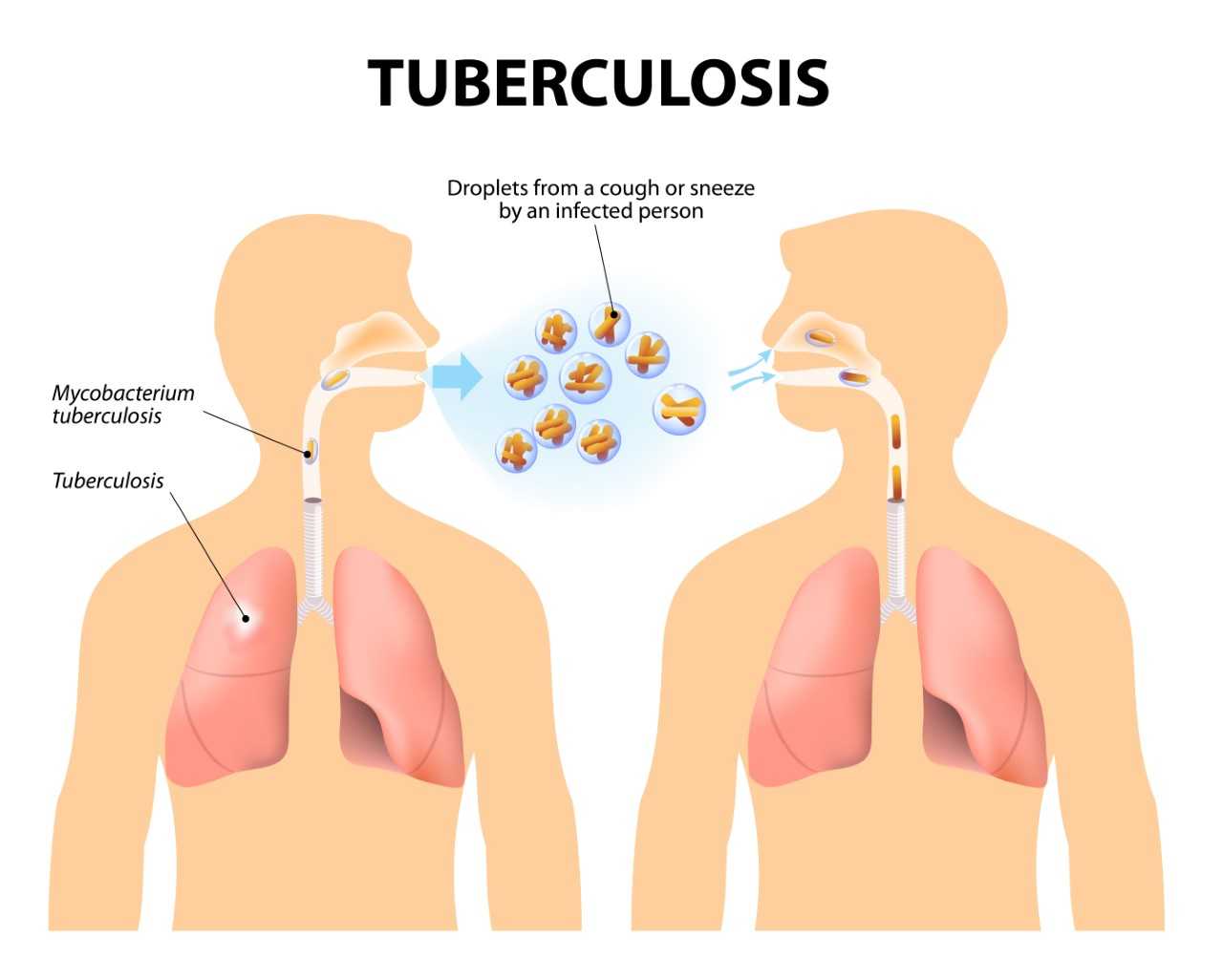
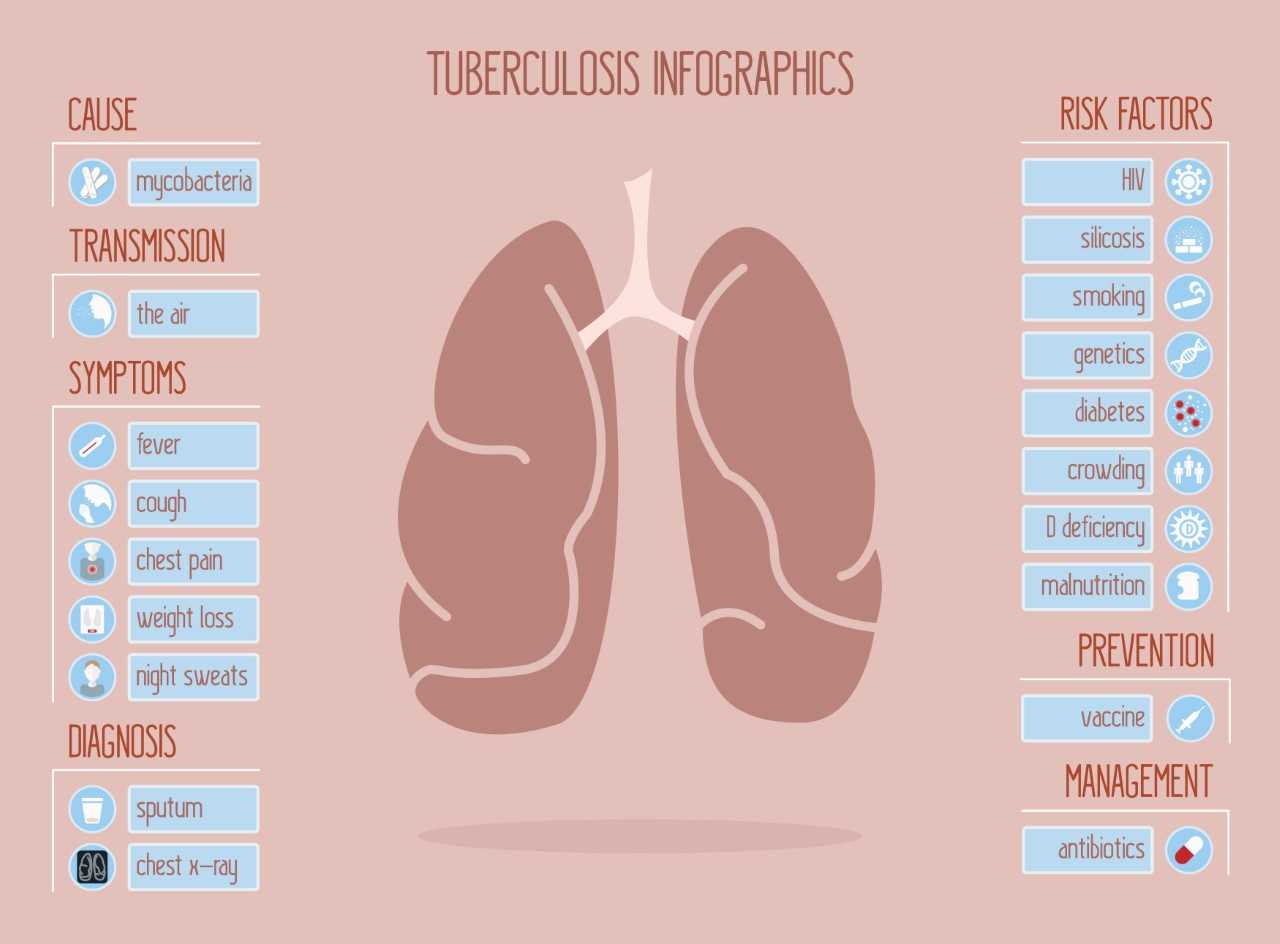
Detect TB at the Right Time with Comprehensive Range of Tests
- 30 Aug, 2017
- Written by Team Dr Lal PathLabs
Medically Approved by Dr. Shuchi
Table of Contents

HAINS ASSAY
A BREAKTHROUGH MULTIPLEX PCR LINE PROBE ASSAY THAT DETECTS MDR TB
• Eliminate Turn Around Time (TAT) from specimen collection to reporting results <7 days compared to a TAT of two to three months for conventional testing.
• Overall performance of this assay is superior to conventional testing (American Journal of Respiratory & Critical Care Medicine).
SPECIMEN
Submit 2mL (1mL min.) Sputum/Bronchoalveolar Lavage/Pleural Aspirates OR Cultured growth on appropriate media in a strile screw capped container in a sealed plastic bag. Ship refrigerated. DO NOT FREEZE.
USAGE
Detecting M. tuberculosis complex and specific mutations in the rpoB gene conferring rifampicin resistance and specific mutations in the katG gene conferring high-level isoniazid resistance, as well as those in the inhA gene conferring low-level isoniazid resistance.
AFB CULTURE
CULTURE, AFB (IMYCOBACTERIUM), RAPID DOES NOT INCLUDE ANT1-TUBERCULAR DRUG SENSITIVITY
• Ensures 100% commensal free culture-PANTA EDGE
• Rapid-Results within 2-4 weeks
• Highly sensitive- Can detect 10 viable bacillt/mL
• Differentiates typical and atypical mycobacteria with speciation offered by MALDI-TOF
• Culture stored for 2 weeks for ordering drug sensitivity
• Special culture vials for AFB blood culture (Myco F-Lytic) to improve detection
GENXPERT
*GENEXPERT MTB WITH REFAMPICIN, QUALITATIVE, PCR
SPECIMEN
• 3mL (1mL min.) Sputum OR 3mL (1 mL mini,) concentrated sediments prepared from induced or expectorated sputum OR 3mL (1 mL.min.) BAL fluid OR 2mL (1mL min.)CSF OR 5mL (3mL min.) Body fluids/Aspirates OR Pus in a sterile screw capped container. Ship refrigerated (DO NOT FREEZE).
• To detect Mycobacterium tuberculosis complex and to elucidate drug resistance to Rifampicin.
GENEXPERT.COM
*GENEXPERT WITH RIFAMPICIN RESISTANCE, CULTURE AFB RAPID
SPECIMEN
6mL (3mL mini) first morning Sputum OR 6mL (3mL mini.) concentrated sediments prepared from induced or expectorate Sputum OR 6mL (3mL mini.) BAL fluid OR 2mL (lmL mini.) CSF OR 6mL (3m1 mini.) BAL fluid OR 2mL (lmL min.) CSF OR 6mL (3mL min.) Body fluids /Aspirates OR 2mL (lmL min.) Pus in a sterile screw capped container. Ship refrigerated (DO NOT FREEZE).
USAGE
To detect Mycobacterium tuberculosis complex and to elucidate drug resistance to Rifampicin.
In addition, this test differentiates between MTB and MOTT by rapid TB antigen MPT64.
GENEXPERT REFLEX TESTING WITH 4 DRUG SENSITIVITY
*GENEXPERT WITH RIFAMPICIN RESISTANCE, CULTURE AFB, RAPID WITH ANTITUBERCULAR SENSITIVITY TO STREPTOMYCIN, ISONIAZID, ETHAMBUTOL & PYRAZINAMIDE.
SPECIMEN
Same as Gonexpert.com.
USAGE
To detect Mycobacterium tuberculosis complex in pulmonary samples and to elucidate drugresistance to Anti-tubercular drugs.
In addition, this test differentiates between MTB and MOTT by rapid TB Antigen MPT64.
TB.COM
MYCOBACTERIUM COMBINED PANEL: TB.COM
*MYCOBACTERIUM TUBERCULOSIS, PCR *CULTURE, AFB RAPID *ZIEHL NEELSON STAIN
SPECIMEN
First morning Sputum on 3 consecutive days or 5-10mL Body fluids/aspirate OR 2mL CSF or 100mL first morning Urine collection on 3 consecutive days OR fresh tissue biopsies including endometrial aspirate/curettage in sterile normal saline OR 1-5mL_ Whool Blood/Bone Marrow in 1 Lavender Top (EDTA)/Yellow Top (ACD) tube OR 1-2mL Menstrual blood in a sterile screw capped container AND 3mL (1mL min.) blood in Myco F/ Lytic bottle available from LPL. All specimens to be collected aseptically and shipped refrigerated.
USAGE
This is the recommended test for diagnosis of Mycobacterial infections.
MYCOSURE
THE ONE TEST THAT ELIMINATES CHANCES OF FAILURE IN TB THERAPY
NEW MYCOSURE (MYCOBACTERIUM TUBERCULOSIS PCR) FOR DETECTION OF MTB AND DRUG RESISTANCE,
• Drug resistance to 4 drugs- Rifampicin, Isoniazid. Ethambutol and Pyrazinamide elucidated by molecular typing of TB bacteria
• Unique set of primers enhance sensitivity and specificity of assay
• Semi-quantitative assay- positivity reported as high, medium, low or very low
SPECIMEN
Submit first morning Sputum on 3 consecutive days OR 5-10mL Body fluids/Aspirates OR 2mL CSF, OR 100mL (50mL min.) first morning Urine collected on 3 consecutive days OR Fresh tissue biopsies including endometrial aspirate curettage in sterile normal saline OR Formalin fixed paraffin embedded Tissue block OR 3mL (1mL min.) Whole Blood/Bone Marrow in 1 Lavender Top (EDTA)/Yellow Top (ACD) tube OR Menstrual Mood in a sterile screw capped container OR culture isolates. All specimens to be collected aseptically and shipped refrigerated. DO NOT FREEZE.
DR LAL PATHLABS ADVANTAGE
• 2 targets for detection of MTB complex
• IS6110 multi-copy gene for increased sensitivity & MPB64 to compensate for strains lacking IS6110
• 1 target for detection of NTM
• Inhibition detected by addition of internal control to every sample
USAGE
• This is a sensitive PCR assay for the detection of Mycobacterium tuberculosis complex and Non-tuberculosis Mycobacteria (NTM).
TB PLATINUM
SPECIMEN
5 mL (3 mL min.) whole blood from 1 Green top (Lithium heparin) tube. Ship refrigerated.
• TB Platinum is based on the principle of Interferon Gamma Release Assay (IGRA).
• Based on quantitative detection of Interferon Gamma (FN-a) that is released by in-vitro stimulation by Mycobacterium tuberculosis antigens.
• Now at more economical cost.
QUANTIFERON TB GOLD
Latent Tuberculosis Infection (LTB) is a no communicable asymptomatic condition that persists for many years and may progress to Tuberculous disease. The main aim of diagnosing LTBI is to consider medical treatment for preventing the disease. This test is a measure of cell mediated immune response to antigens simulating the mycobacterial proteins. A positive result indicates that mycobacterium tuberculosis infection is likely but further medical and diagnostic evaluation is necessary. This test is usually negative in individuals vaccinated with Mycobacterium bovis BCG.
SPECIMEN
Collect 1 mL whole blood each in a set of special Quantiferon TB Gold tubes available from LPL. Shake the tubes vigorously at Least 10 times to ensure thorough mixing. DO NOT CENTRIFUGE. Ship at room temperature to reach lab within 16 hours OR incubate the tubes immediately at 37°C for 16-24 hours and centrifuge. Ship refrigerated. DO NOT FREEZE.